Lessons learned from Optima Belle
On Tuesday December 8, 2020, at approximately 10 pm EST an explosion occurred at Optima Chemical LLC in Belle, WV. The incident lead to one fatality and two injuries as well as a shelter-in-place for community members within a 2-mile radius of the facility.
A pressure-rated rotary double cone dryer containing a chlorinated isocyanurate compound (sodium dichloroisocyanurate dihydrate, or NaDCC dihydrate, trade named CDB-56®) exploded, causing a subsequent fire and toxic chlorine release at Optima Belle LLC (Optima Belle) in Belle, West Virginia.
Optima Belle’s estimated property damage from the incident is $33.1 million.
The chemical safety board in the USA recently released its report into the incident. One of the report’s recommendations is the use of an assessment processes called the O.R.E.O.S method to assess whether a chemical presents a risk before it is used in scale up / synthesis work. O.R.E.O.S was first outlined in a paper published in 2021 by Vertex Pharmaceutials and Sperry et al.
It works by assesing a number of factors about a chemical including the presence of explosive functional groups, oxygen balance, rule of 6, onset temperatures, yoshida correlations and scale.
This methodology, amongst others is incorporated in our DG Assessor tool, which we believe it’s the only commercially available tool to do this. Had it been available in 2020 it would have instantly and correctly identified the chemicals involved as being at high risk of an explosion (see below).
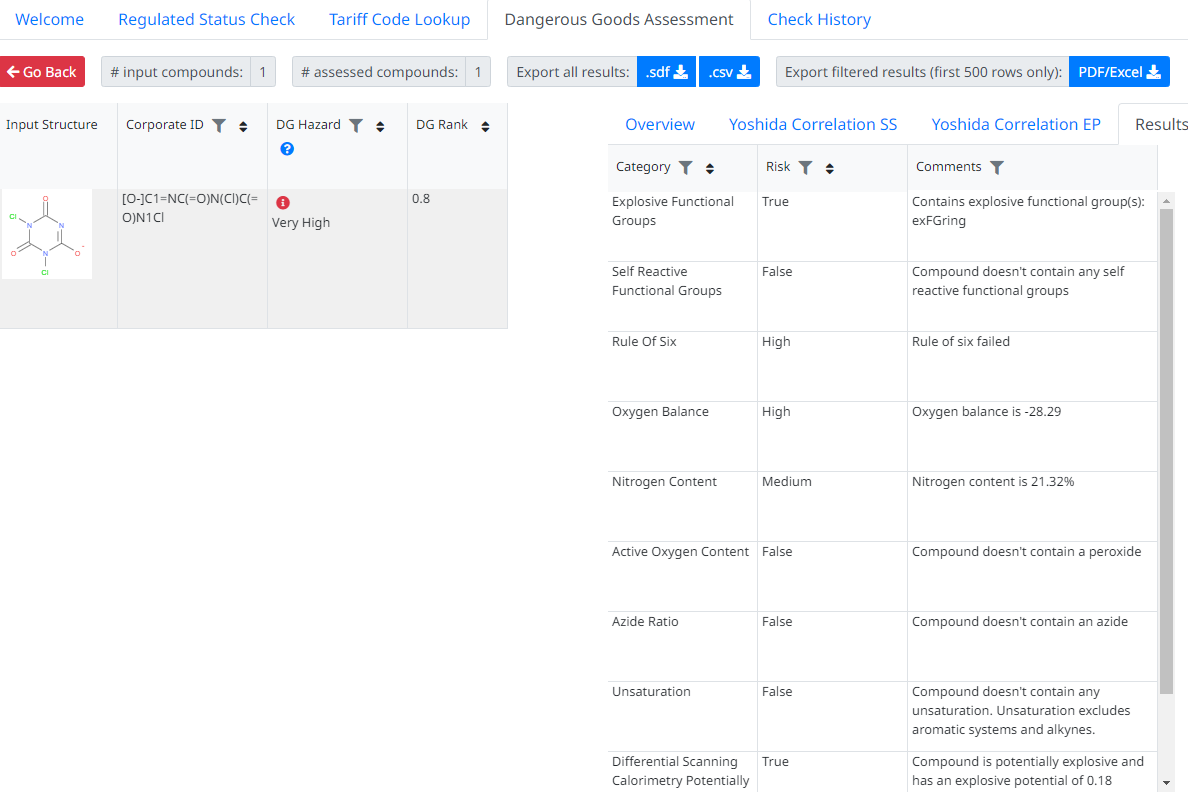
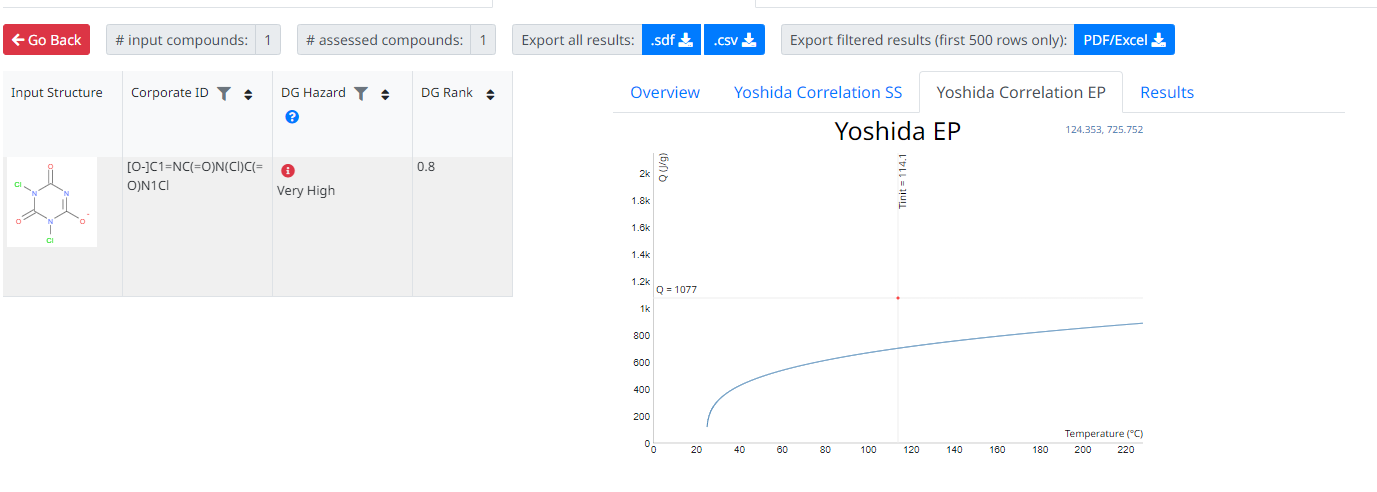
The example above includes the output (Tinit and energy released) from a DSC (Differential Scanning Calorimetry) instrument, allowing DG Asssesor to also calculate Yoshida correlations and take these into account. However, DSC data is often not always available and even without this, working purely on the chemical structure, DG Asssesor still classified this as a high risk chemical with regards to explosive / energetic release.
Developed in conjunction with a major Pharmaceutical companies safety and process chemistry groups. Its algorithms and checks also help fulfil the requirements and assessments laid out in the UN Manual of Tests and Criteria for assessing chemicals for potential safety concerns before handling or shipping and ECHA CLP assesment procedures (section 2.1.4) 'Screening procedures and waiving of testing' for chemicals.
If you would like to learn more about this an DG Assessor, please contact us.
Trusted by our Clients











